More Information
Submitted: October 03, 2025 | Accepted: March 13, 2026 | Published: March 16, 2026
Citation: El Athmani O, El Ansari N, Isfaoun Z, Lakhrissi M, El Kababri M, Kili A, et al. Diagnostic Pitfall Thoraco-abdominal Neuroblastoma Initially Misdiagnosed as Pulmonary Tuberculosis. J Adv Pediatr Child Health. 2026; 9(1): 001-004. Available from:
https://dx.doi.org/10.29328/journal.japch.1001079
DOI: 10.29328/journal.japch.1001079
Copyright License: © 2026 El Athmani O, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Child; Neuroblastoma; Thoracoabdominal mass; Tuberculosis misdiagnosis; Tumor
Diagnostic Pitfall Thoraco-abdominal Neuroblastoma Initially Misdiagnosed as Pulmonary Tuberculosis
Omayma El Athmani1*, Naoual El Ansari2, Zineb Isfaoun2, Mariame Lakhrissi2, Maria El Kababri2, Amina Kili2, Mohamed El Khorassani2 and Laila Hessissen2
1Pediatrics Department, Children’s Hospital, University Hospital Center Ibn Sina, Faculty of Medicine and Pharmacy, Rabat, MAR, Morocco
2Pediatric Hemato-Oncology Department, Children’s Hospital, University Hospital Center Ibn Sina, Faculty of Medicine and Pharmacy, Mohammed V University, Rabat, MAR, Morocco
*Corresponding author: Omayma El Athmani, Pediatrics Department, Children’s Hospital, University Hospital Center Ibn Sina, Faculty of Medicine and Pharmacy, Rabat, MAR, Morocco, Email: [email protected]
Neuroblastoma is the most common extracranial solid malignancy of childhood and typically arises from neural crest cells of the sympathetic nervous system, most frequently in the adrenal medulla or paravertebral sympathetic chain. Thoracic or thoracoabdominal localization is less common and may present with nonspecific respiratory symptoms that mimic infectious diseases. We report the case of a three-year-old boy presenting with prolonged fever, persistent cough, and progressive weight loss. Initial chest radiography and pleural fluid analysis suggested pulmonary tuberculosis, and antituberculous therapy was initiated.
However, the absence of clinical improvement prompted further evaluation. Contrast-enhanced thoracoabdominal computed tomography revealed a large prevertebral mass extending into the posterior mediastinum. Ultrasound-guided biopsy followed by histopathological and immunohistochemical examination confirmed the diagnosis of neuroblastoma. Staging investigations demonstrated bone and bone marrow involvement consistent with high-risk disease. The patient was treated according to the NBL-MA- 2010 protocol with significant clinical improvement and is currently scheduled for consolidation with autologous stem cell transplantation. This case highlights an important diagnostic pitfall in tuberculosis- endemic regions and emphasizes the importance of early cross-sectional imaging and tissue diagnosis in children presenting with persistent thoracic pathology.
Categories: Oncology, Pediatrics
Neuroblastoma (NB) is a malignant pediatric tumor arising from primitive neural crest cells that form the sympathetic nervous system. This embryological origin explains its frequent occurrence in the adrenal medulla and along the paravertebral sympathetic chain [1,2].
Neuroblastoma is the most common extracranial solid tumor of childhood and accounts for approximately 8% - 10% of all pediatric cancers. The disease predominantly affects infants and young children, with the highest incidence occurring during the first year of life [3,4].
Historically, the first descriptions of neuroblastoma date back to the early twentieth century. Thoracic localizations were subsequently described several decades later as imaging techniques improved and mediastinal tumors became better characterized [5,6].
The abdomen represents the most common primary site, accounting for approximately 75% of cases. Thoracic neuroblastoma represents a smaller proportion of cases and has been reported in approximately 11% - 26% of patients [3].
Clinical manifestations of thoracic neuroblastoma are often nonspecific and may include cough, dyspnea, fever, or chest discomfort. Because these symptoms overlap with common respiratory infections, diagnosis may be delayed, particularly in regions where pulmonary tuberculosis remains prevalent [7,8].
In tuberculosis-endemic areas, radiographic findings such as pulmonary opacity or pleural effusion may initially suggest an infectious etiology. Several reports have described cases of thoracic neuroblastoma initially treated as pulmonary infection before cross-sectional imaging and histopathological confirmation established the correct diagnosis [9,10].
In this report, we describe a case of thoracoabdominal neuroblastoma initially misdiagnosed as pulmonary tuberculosis, highlighting the diagnostic challenges encountered in tuberculosis-endemic regions.
A three-year-old boy, the second child of non-consanguineous parents, presented in December 2023 with prolonged fever, persistent cough, and progressive weight loss. Initial empirical antibiotic therapy with amoxicillin failed to improve the clinical condition.
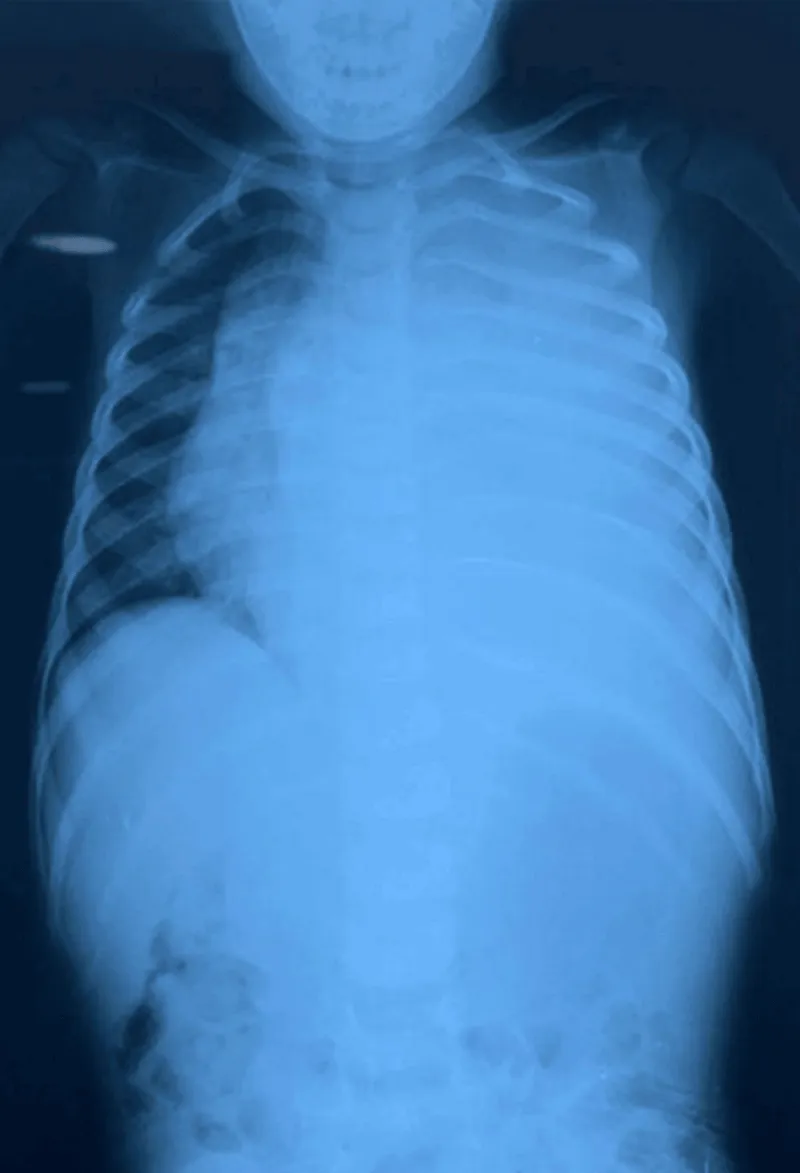
A chest radiograph performed in January 2024 demonstrated a large homogeneous opacity occupying the left hemithorax with mediastinal shift toward the contralateral side (Figure 1).
Figure 1: Chest Radiograph at Initial Presentation.
Chest radiograph demonstrating a large homogeneous left-sided thoracic opacity associated with mediastinal shift to the contralateral side, initially suggestive of pulmonary infection.
Diagnostic thoracentesis was performed. Pleural fluid analysis revealed an exudative effusion with lymphocyte predominance. The results of the pleural fluid analysis are summarized in Table 1. Based on these findings and the high prevalence of tuberculosis in the region, a presumptive diagnosis of pulmonary tuberculosis was made and antituberculous therapy was initiated.
| Table 1: Pleural Fluid Analysis. | ||
| Parameter | Result | Interpretation |
| Appearance | Turbid yellow fluid | Suggestive of exudate |
| Protein | 4.1 g/dL | Elevated |
| LDH | 650 U/L | Elevated |
| Glucose | 80 mg/dL | Within normal range |
| Total cell count | 1,900 cells/mm³ | Increased |
| Differential count | Lymphocytes 70%, Neutrophils 20%, Macrophages 10% | Lymphocyte predominant |
| Cytology | No malignant cells detected | Non-diagnostic |
Pleural fluid obtained during diagnostic thoracentesis showed an exudative effusion with lymphocyte predominance. Cytological examination did not reveal malignant cells.
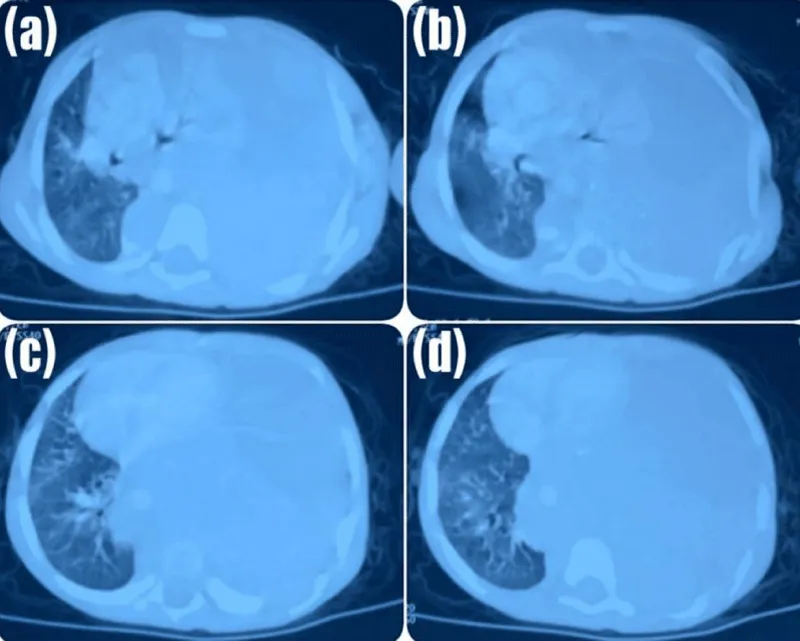
Despite treatment, the patient developed progressive respiratory distress. Contrast-enhanced thoracoabdominal computed tomography revealed a large prevertebral mass extending into the posterior mediastinum (Figure 2), raising strong suspicion for neuroblastoma.
Figure 2: Contrast-Enhanced Thoraco-Abdominal CT Scan.
(a) Axial CT section showing a large heterogeneous soft-tissue mass occupying the majority of the hemithorax and causing marked compression of the adjacent lung parenchyma. Only a small portion of aerated lung is visible laterally.
(b) Slightly inferior section demonstrating persistence of the mass with displacement of mediastinal structures and further compression of the ipsilateral lung.
(c) Lower thoracic section showing the inferior extension of the lesion, with partial visualization of compressed lung parenchyma and heterogeneous internal density of the mass.
(d) Most inferior section illustrating the continued presence of the mass with significant reduction of the aerated lung field and pronounced mass effect on surrounding thoracic structures.
Ultrasound-guided core biopsy was subsequently performed. Histopathological examination demonstrated a tumor composed of small round cells with hyperchromatic nuclei embedded in a fibrillary background, consistent with neuroblastoma. Immunohistochemical analysis supported the diagnosis. Molecular analysis indicated the absence of MYCN amplification.
Staging investigations revealed bone and bone marrow involvement, classifying the disease as high-risk neuroblastoma. The patient was started on chemotherapy according to the NBL-MA-2010 protocol. Clinical improvement was observed after the first cycle, particularly with significant reduction in respiratory symptoms. The patient is currently undergoing further oncological management and is scheduled for consolidation with autologous stem cell transplantation.
The clinical presentation of neuroblastoma varies considerably depending on the location of the primary tumor and the extent of metastatic disease. Neuroblastoma is a malignant pediatric tumor arising from neural crest cells and represents the most common extracranial solid tumor of childhood [1,2].
Thoracic neuroblastoma represents a less common presentation of the disease but may produce respiratory symptoms that mimic common pulmonary conditions. Patients may present with cough, dyspnea, chest pain, or fever due to mediastinal compression by the tumor [3]. In clinical series of thoracic neuroblastoma, respiratory manifestations have been reported as the most frequent presenting symptoms.
Neuroblastoma is also characterized by marked biological heterogeneity, ranging from spontaneous regression in infants to aggressive metastatic disease in older children [7]. Such variability frequently contributes to delayed diagnosis or misdiagnosis.
Less typical manifestations have also been described. These include persistent vomiting in neonates and thoracic skeletal abnormalities such as pectus excavatum associated with mediastinal tumors [11,12]. In addition, thoracic neuroblastoma may occasionally present with neurological symptoms related to spinal cord compression when the tumor extends through the intervertebral foramina.
In tuberculosis-endemic regions, these clinical and radiological findings may easily lead to an initial diagnosis of pulmonary tuberculosis. In the present case, the presence of lymphocyte-predominant pleural effusion and thoracic opacity on chest radiography initially supported this diagnosis. However, the absence of clinical improvement under antituberculous therapy prompted further investigation and ultimately led to the identification of a thoracoabdominal mass.
Imaging plays a central role in the diagnostic evaluation of neuroblastoma. Cross-sectional imaging techniques such as computed tomography and magnetic resonance imaging allow accurate assessment of tumor location, local invasion, and metastatic spread [13]. Functional imaging with metaiodobenzylguanidine scintigraphy is particularly valuable because approximately 90% of neuroblastomas demonstrate avid tracer uptake [14].
Two major staging systems are currently used to evaluate disease extent and guide prognosis. The International Neuroblastoma Risk Group Staging System allows pretreatment risk stratification based on imaging-defined risk factors independent of surgical intervention [7]. The International Neuroblastoma Staging System categorizes tumors according to surgical findings and the extent of resection [15].
Several studies have suggested that thoracic neuroblastomas may be associated with relatively more favorable outcomes compared with tumors arising at other anatomical sites [16]. However, treatment strategies remain based on established risk-adapted protocols.
Management of high-risk neuroblastoma requires multimodal therapy combining intensive chemotherapy, surgical resection when feasible, radiotherapy, and consolidation with high-dose chemotherapy followed by autologous stem cell transplantation [17]. In Morocco, treatment protocols such as the NBL-MA-2010 protocol have been developed to adapt international therapeutic strategies to local healthcare resources while maintaining effective oncological management [18].
Thoracoabdominal neuroblastoma may present with nonspecific respiratory manifestations and mimic pulmonary infections such as tuberculosis, particularly in endemic regions. This diagnostic overlap may lead to delayed recognition and treatment. Clinicians should maintain a high index of suspicion for malignant etiologies in children presenting with persistent thoracic pathology that does not respond to antimicrobial therapy. Early cross-sectional imaging and prompt histopathological confirmation are essential for accurate diagnosis and timely initiation of appropriate risk-adapted treatment.
- Park JR, Eggert A, Caron H. Neuroblastoma: biology, prognosis, and treatment. Pediatr Clin North Am. 2015;62:225-46. Available from: https://doi.org/10.1016/j.hoc.2009.11.011
- Brodeur GM. Neuroblastoma: biological insights into a clinical enigma. Nat Rev Cancer. 2003;3:203-16. Available from: https://doi.org/10.1038/nrc1014
- Demir HA, Yalçin B, Büyükpamukçu N, Kale G, Varan A, Akyüz C, et al. Thoracic neuroblastic tumors in childhood. Pediatr Blood Cancer. 2010;54:885-9. Available from: https://doi.org/10.1002/pbc.22405
- Yan P, Qi F, Bian L, Xu Y, Zhou J, Hu J, et al. Comparison of incidence and outcomes of neuroblastoma in children, adolescents, and adults in the United States: a Surveillance, Epidemiology, and End Results (SEER) program population study. Med Sci Monit. 2020;26:927218. Available from: https://doi.org/10.12659/MSM.927218 Available from: https://doi.org/10.12659/MSM.927218
- Goon HK, Cohen DH, Harvey JG. Review of thoracic neuroblastoma. Aust Paediatr J. 1984;20:17-21. Available from: https://doi.org/10.1111/j.1440-1754.1984.tb00030.x
- Rubie H, Hartmann O, Giron A, Lemoine G, Gruner M, Brugieres L, et al. Nonmetastatic thoracic neuroblastomas: a review of 40 cases. Med Pediatr Oncol. 1991;19:253-7. Available from: https://doi.org/10.1002/mpo.2950190408
- Cohn SL, Pearson ADJ, London WB, Monclair T, Ambros PF, Brodeur GM, et al. The International Neuroblastoma Risk Group (INRG) classification system. J Clin Oncol. 2009;27:289-97. Available from: https://doi.org/10.1200/jco.2008.16.6785
- Caron HN. Are thoracic neuroblastomas really different?. Pediatr Blood Cancer. 2010;54:867. Available from: https://doi.org/10.1002/pbc.22470
- Rezaimehr Y, Bhargava R. Neuroblastoma presenting as persistent postprandial emesis in a neonate. Pediatr Emerg Care. 2013;29:1273-5. Available from: https://doi.org/10.1097/PEC.0000000000000029
- Yoh T, Ohnuki T, Ishikura T, Kei J, Obara T, Nitta S. A rare case of mediastinal neuroblastoma with pectus excavatum in a 4-month-old boy: a surgical report. Nihon Kyobu Geka Gakkai Zasshi. 1992;40:2227-31. Available from: https://pubmed.ncbi.nlm.nih.gov/1491204/
- Yiin JJ, Chang CS, Jan YJ, Wang WC. Treatment of neuroblastoma with intraspinal extensions. J Clin Neurosci. 2003;10:579-83. Available from: https://doi.org/10.1016/S0967-5868(03)00064-X
- Morris JA, Schochat SJ, Smith EI, Look AT, Brodeur GM, Cantor AB, et al. Biological variables in thoracic neuroblastoma: a Pediatric Oncology Group study. J Pediatr Surg. 1995;30:296-303. Available from: https://doi.org/10.1016/0022-3468(95)90577-4
- Ahmed AA, Zhang L, Reddivalla N, Hetherington M. Neuroblastoma in children: update on clinicopathologic and genetic prognostic factors. Pediatr Hematol Oncol. 2017;34:165-85. Available from: https://doi.org/10.1080/08880018.2017.1330375
- Olecki E, Grant CN. MIBG in neuroblastoma diagnosis and treatment. Semin Pediatr Surg. 2019;28:150859. Available from: https://doi.org/10.1016/j.sempedsurg.2019.150859
- Häberle B, Hero B, Berthold F, von Schweinitz D. Characteristics and outcome of thoracic neuroblastoma. Eur J Pediatr Surg. 2002;12:145-50. Available from: https://doi.org/10.1055/s-2002-32721
- Castleberry RP, Shuster JJ, Smith EI. The Pediatric Oncology Group experience with the International Staging System criteria for neuroblastoma. J Clin Oncol. 1994;12:2378-81. Available from: https://doi.org/10.1200/JCO.1994.12.11.2378
- Yoneda A. Role of surgery in neuroblastoma. Pediatr Surg Int. 2023;39:177. Available from: https://doi.org/10.1007/s00383-023-05459-1
- Salman Z, El Kababri M, Hessissen L, Khattab M, Matthay K. An intensive induction protocol for high-risk neuroblastoma in Morocco. J Glob Oncol. 2016;2:80. Available from: https://doi.org/10.1200/JGO.2016.004259